Sabina Paglialunga, PhD & Aernout van Haarst, PhD, Senior Directors, Scientific Affairs, Celerion
Cocktail drug-drug interaction (DDI) studies are an efficient and effective approach to simultaneously evaluate the effect of a study drug on multiple CYP450 enzymes and transporter substrates at once. Advantages of this approach include a potential reduction in the number of DDI studies, overall shortening of clinical study timelines, cost savings and faster data access. At Celerion, we regularly run cocktail DDI trials, with over a dozen studies completed in the past 3 years.
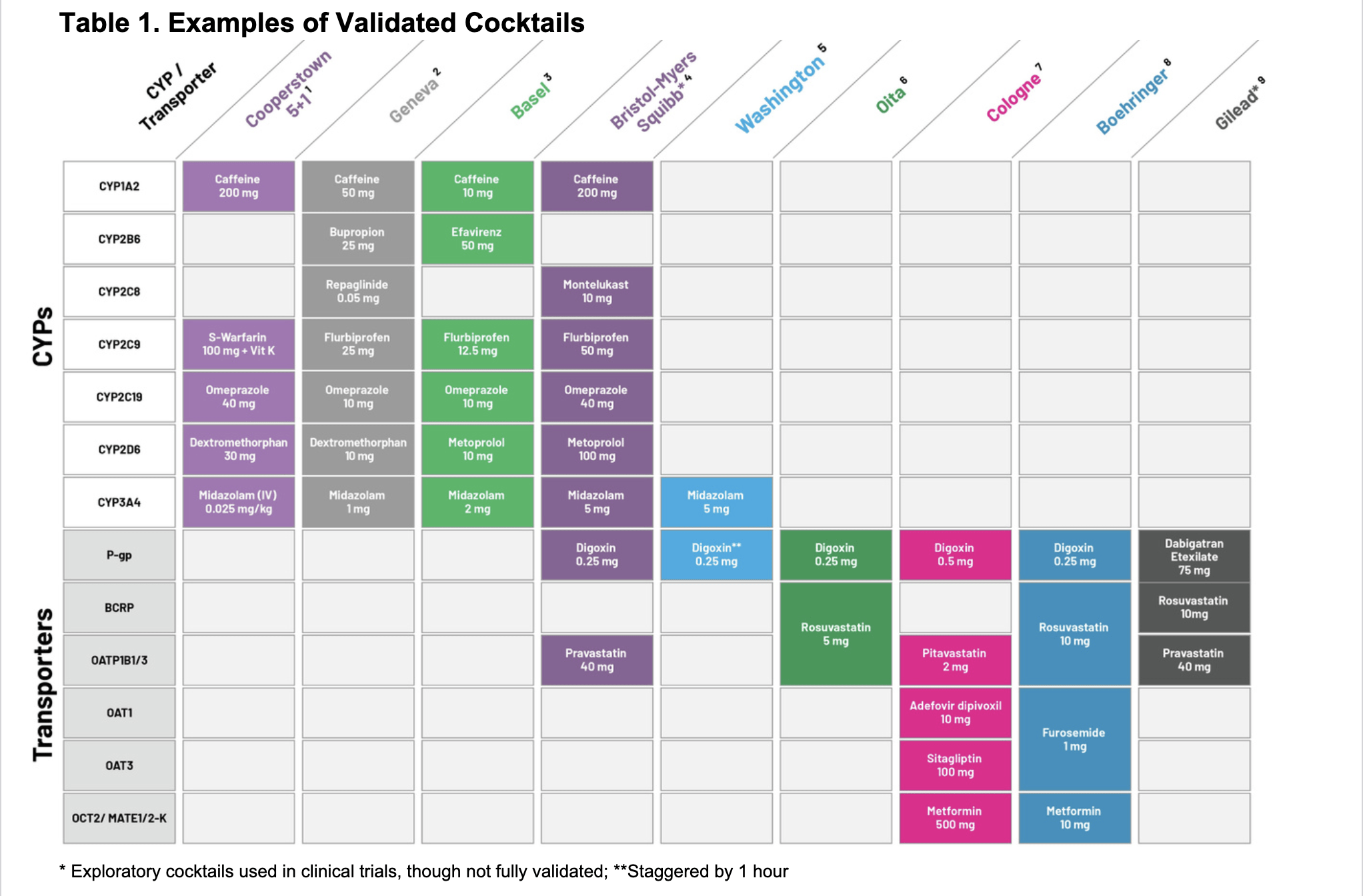
Well-known and popular substrate cocktails for CYPs and transporters include:
While substrate cocktails were first introduced in the FDA DDI guidance in 2006 [6] as an option for drug developers, some sponsors may be hesitant with applying this approach. In this blog article, we break down 5 common myths surrounding cocktail DDI studies, and provide facts to support this efficient strategy:
5 Common Myths About Cocktail DDI Studies:
Myth #1: It is not safe to combine all these drugs.
FACT: Validated substrate cocktails have undergone rigorous safety testing in a healthy volunteer population. At the given drug doses tested, they have been found safe to combine in a cocktail.
Myth #2: There may be PK interactions among the probe drugs.
FACT: In addition to safety assessments, validated cocktails were shown not to interact from a PK perspective at dose levels relevant to the clinical dose, nor to interfere with mutual bioanalytical assays. Each substrate in the cocktail was assessed alone and in combination with the other probe drugs during the validation process. For example, in the Cooperstown 5+1 cocktail, which combines caffeine (CYP1A2 substrate), warfarin (CYP2C9 substrate) + vitamin K, omeprazole (CYP2C19 substrate), dextromethorphan (CYP2D6 substrate) and midazolam (CYP3A substrate), PK and safety of the cocktail were evaluated for each component alone and as a cocktail.
Myth #3: Regulatory agencies won’t accept cocktail DDI data.
FACT: The FDA has been encouraging use of substrate cocktails since 2006 [10], and this recommendation is reiterated in the most recent ICH M12 guidance published in 2024. Moreover, based on our internal records, approximately 25% of approved new molecular entities (NME) have employed a cocktail DDI during development.
Myth #4: Validated cocktails don’t address all the CYPs / transporters of interest for my study drug.
FACT: If there is no validated cocktail available covering all the CYP’s and transporters that are relevant for a particular new drug, we encourage a staggered/sequential design approach. In such a design, sequential administration of several (cocktails of) substrates can be incorporated into a single DDI arm. As many substrates have short half-lives, sequential administration may only add a couple of extra days to overall study conduct.
Myth #5: The total blood volume will be too high.
FACT: When applying a cocktail DDI approach, blood samples need to be collected for bioanalysis of each individual probe. The total blood collection volume is therefore higher than typically observed in a single-substrate DDI study. However, in our experience the total blood volume collected in cocktail DDI studies in general remains well below the recommended maximum collection limit of 500 mL, which in part can be attributed to our validated bioanalytical substrate assays being optimized to use the smallest blood sample volume to help keep total blood collection volume to a minimum. Particularly in cases where blood collection volume may reach a critical value, development and validation of combined assays for multiple probes may offer a means to further reduce the volume of blood draws.
Cheers to the Cocktails!
In summary, the use of validated substrate cocktails in clinical DDI studies is well supported by both regulatory agencies and a robust body of scientific evidence. Common concerns regarding safety, PK interactions, regulatory acceptance, comprehensiveness of CYP/transporters coverage, and blood volume requirements can be thoroughly addressed through careful study design, validation of cocktails and ongoing advancements in bioanalytical methodologies. As a result, cocktail approaches have become a trusted and efficient strategy to evaluate metabolic pathways, streamline clinical development, and ultimately enhance the safety of both trial participants and target patient populations.
Our expert team of PK scientists can help optimize your next substrate DDI study with the thoughtful and efficient application of cocktails. Cheers!
References:
-
- Chainuvati et al. (2003) CPT
- Aurinsalo et al. (2025) CPT
- Suenderhauf et al. (2020) BJCP
- Tye et al. (2016) Int J Pharm
- Kirby et al. (2006) JCP
-
- Otani et al. (2019) CTS
- Trueck et al. (2019) CPT
- Wiebe et al. (2020) Clin PK
- Younis et al. (2023) Clin PK
- FDA (2006) Drug Interaction Studies — Study Design, Data Analysis, and Implications for Dosing and Labeling. https://downloads.regulations.gov/FDA-2006-D-0036-0002/attachment_1.pdf
- ICH (2024). Drug Interaction Studies M12. https://database.ich.org/sites/default/files/ICH_M12_Step4_Guideline_2024_0521_0.pdf
