COVID-19 is a highly infectious respiratory disease caused by the SARS-CoV-2 virus that has affected every corner of the world and nearly all aspects of daily life. In a subset of COVID-19 patients, an exaggerated immune response can lead to acute respiratory distress syndrome (ARDS) requiring mechanical ventilation and leading to death. For society to return to “normal”, it is estimated that ~70% of herd immunity is required [1], which could result in thousands of casualties. Furthermore, this herd immunity target may not be achievable because it is still unknown if those with mild or asymptomatic cases of COVID-19 may not have built up sufficient immunity to prevent reinfection. Therefore, we must consider vaccination as the only viable option to eliminate this virus and thus the race is on to develop an effective vaccine against SARS-CoV-2.
Under the Microscope:
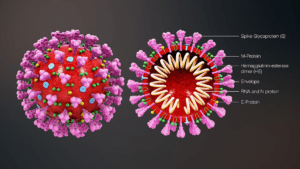
SARS-CoV-2 is a single stranded RNA virus, it is encapsulated by proteins and lipids. The virus has four main structural proteins; a spike glycoprotein (S), a small envelope glycoprotein (E), a membrane glycoprotein (M) and a nucleocapsid protein (N) in addition to other accessory proteins. A homotrimer of S-proteins facilitates binding to angiotensin-converting enzyme 2 (ACE2) receptor on host cells and cell entry [2]. The S-protein is therefore a key site for antibody neutralization, but vaccines developed against other viral protein are also under investigation.
Vaccines 101:
Vaccines boost immunity against infectious diseases through controlled exposure of an antigen, which can be an attenuated virus or fragments of viral proteins. The immune system responds by generating antibodies that protect against future infection. Subsequent exposure to the virus or another infected individual then triggers antibody recognition and the virus is cleared via the immune system activation. Adjuvants are applied to vaccine formulation to upregulate the antigenic response, and depending on the duration of protection, an additional booster shot may be needed.
Vaccine Safety Assessments:
Early clinical phase studies focus on safety, tolerability and immunogenicity, and throughout vaccine development particular attention is paid to hypersensitivity. While vaccines are generally considered safe, serious anaphylactic adverse events associated with immunization can occur, albeit they are extremely rare. Hypersensitivity to the antigen, adjuvants and preservatives have been observed [3] and may require dose adjustment or re-formulation. Another aspect to consider during vaccine development is the potential to induce a Th2 response. Th2 is one of two T-cell responses stimulated when antigens are presented to T cells. The type of T cell response (Th1 vs Th2) results in a particular set of cytokines released. Vaccines depend on a Th1 response to generate immunoglobulins, which elicit immunity against viruses, bacterial and fungal infections. A Th-2 response can counteract Th1 by upregulating interleukin-10 which has anti-inflammatory function [4]. If a Th2-type response is established upon immunization, it can prevent Th1-type response as these are antagonistic processes and a Th2-bias can potentially exacerbate the infection [5].
Bioanalytical Support of Vaccine Trials:
Advanced bioanalytical assays are needed for efficacy and safety measures. It is important that these bioanalytical tests are robust and analytically validated for their context of use in order to support a clinical trial [6]. A variety of assays are available for vaccine drug development, which induce antibody titer, qPCR, ELISpot, cell profiling and cytokine biomarkers.
Table 1. Bioanalytical Assays for Vaccine Development
| Bioanalytical Assays | Utility | Technology |
| Antibody titers | Determine the amount of antibodies produced in response to inoculation | Ligand binding methods such as ELISA or MSD |
| Neutralizing activity of the antibody | Determine the extend in which antibodies can clear the virus | Cell-based assays |
| Viral load | Determine the presences and amount of virus | qPCR |
| DNA vaccines | Quantify exogenous viral DNA components | qPCR |
| mRNA vaccines | Quantify exogenous viral mRNA components | qPCR |
| T-cell profiling | Examine immune cell populations | Flow cytometry |
| T-cell response | Examine IFN-γ activation | ELISpot |
| Antibody producing cells | Evaluate ex-vivo stimulation of PBMC for antibody production | ELISpot |
| Inflammatory cytokines | Monitor the ‘cytokine storm’ and potential risk stratify for disease severity | Ligand binding methods such as ELISA or MSD |
| Neutralizing antibodies (NAbs) assay | Antibodies that develop against the biotherapeutic product that can impact it efficacy or safety profile | Cell-based assays |
| Antibody Dependent Enhancement (ADE) | Characterize the causes of acute lung injury that may occur following coronavirus vaccination | Flow cytometry |
Innovative Vaccine Platforms:
There are several platforms for vaccine development. Each technique holds unique advantages and challenges when it comes to safety, manufacturing and scalability [7-9]. The following explores these considerations for the various vaccines under development during the current pandemic.
- Whole inactivated and live-attenuated virus vaccines: To create a whole inactivated vaccine, the virus is cultured in a laboratory and then killed (inactivated) or weakened (attenuated) with chemicals, heat, or radiation. This process conserves the virus structure, induces neutralizing antibodies and has been applied for other infectious diseases. There is a risk of hypersensitivity and Th2 bias. In the current crisis, a significant amount of live virus would need to be cultured quickly. Viruses are cultured in cell or egg based media, and these vaccines are contraindicated for individuals with an egg allergy. For recombinant live-attenuated vaccines, parts of the genetic sequence of the virus are manipulated to reduce the virulence. The antigens are produced in the body to facility an immune response. There is potential for the virus to revert to a virulent strain therefore this strategy may not be appropriate to inoculate sensitive populations.
- Viral vector vaccines: Weakened adenoviruses or measles viruses are genetically engineered to produce SARS-CoV-2 surface proteins in a patient to elicit an immune response. There is a risk of anti-vector (adenovirus or measles) immunity, lowering the potential immune response against the SARS-CoV-2 target.
- Subunit vaccines: Peptide components or fractions of the surface protein antigens are synthesized to create a vaccine. This strategy has a good safety profile however in most cases, subunit vaccines require adjuvants and booster doses.
- Virus-like particles: The viral outer shell lacking the genetic material is introduced to patients to trigger a strong immune response. By conserving the virus structure, multiple antigens can be displayed. A caveat of this process is that manufacturing on a large scale may be technically challenging.
- Nuclei acid vaccines: DNA or mRNA based vaccines use the patient’s own cell to generate virus peptides and surface proteins that will trigger an immune response (eg. S-protein). The mRNA is encased in a lipid layer that permeates the patients’ cells which act like a bioreactor and transcribe the mRNA into the pathogenic protein that will then stimulate an immune response. DNA vaccines work similarly to mRNA vaccines, with the antigen being coded in a DNA sequence. The DNA is translated to RNA then transcribed to the antigenic peptide. A DNA vaccine does require an extra step to enter into cells, typically with electroporation. One advantage is that more than one viral antigen may be coded. Nucleic acid vaccines are a relatively new technology and have not yet been approved for other infectious diseases.
Table 2. Benefits and Considerations for COVID-19 Vaccine Platforms
| Vaccine Type | Benefits | Considerations |
| Whole inactivated virus | • Conserves viral structure • Rapid development |
• Potential for hyper-sensitivity and Th2-bias • Culturing live virus |
| Live-attenuated recombinant virus | • Site-directed attenuation |
• Potential to revert to virulent strain • Not suitable for sensitive populations |
| Viral vector vaccines | • Robust immune response |
• Cell-based manufacturing • Anti-vector immunity |
| Subunit vaccines | • Good safety profile | • Adjuvant requirement • Manufacturing costs vary |
| Virus-like particles | • Conserves viral structure • Multimeric antigen display |
• Complicated assembly |
| mRNA vaccines |
• Increase the safety profile; non-integrating • Egg and cell free • Rapid, inexpensive and scalable manufacturing |
• May suffer from instability • Low immunogenicity |
| DNA vaccines |
• Non-infectious • Egg and cell free • Greater stability • Potential for multiple antigens • Rapid production |
• Specialized delivery system required • Potential for integration into human genome • Low immunogenicity |
Adapted from: Zhang et al. 2019 [8] & Prompetchara et al. 2020 [9].
A Coordinated Effort:
The urgency of developing, validating and disseminating a COVID-19 vaccine is palpable. Nearly 100* sponsors have shifted resources and pivoted to the COVID-19 indication. Regulatory agencies are even cutting red-tape to expedite clinical trials. The MHRA approved a COVID-19 vaccine trial in 7 working days. In addition, regulatory authorities have also been swift to implement a series of guidance documents to support sponsors and CROs. Recently, the FDA issued guidance on development and licensure of vaccines to prevent COVID-19. In Europe, similar guidelines have been released by EMA and MHRA. Moreover, countries worldwide are ramping up production of syringes, vials and related paraphernalia needed to inoculate millions once a vaccines is approved. This level of swift global coordination has not been seen before.
Ending the Pandemic:
To end the COVID-19 health crisis quickly, we’ll need more than one solution. Biotech and pharma sectors as well as regulatory agencies and CRO stakeholders are up to the challenge as the race for a vaccine is well underway. Promising early results from vaccine developers such as the University of Oxford with a viral vector vaccine, and Moderna with an mRNA vaccine, are hopeful signs that relief is on its way. With a number of different types of vaccines under investigation, this increases our chances of developing several safe and effective COVID-19 vaccines.
References:
1. Randolph HE, Barreiro LB. Herd Immunity: Understanding COVID-19. Immunity. 2020;52(5):737-41.
2. Walls AC, Park YJ, Tortorici MA, Wall A, McGuire AT, Veesler D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell. 2020;181(2):281-92 e6.
3. McNeil MM, DeStefano F. Vaccine-associated hypersensitivity. J Allergy Clin Immunol. 2018;141(2):463-72.
4. Berger A. Th1 and Th2 responses: what are they? BMJ. 2000;321(7258):424.
5. Rosenthal KS, Zimmerman DH. Vaccines: all things considered. Clin Vaccine Immunol. 2006;13(8):821-9.
6. Kar S, Islam R. Rapid and robust bioanalytical assays are critical for SARS-CoV-2 therapeutic and vaccine development and beyond. Bioanalysis. 2020.
7. Thanh Le T, Andreadakis Z, Kumar A, Gomez Roman R, Tollefsen S, Saville M et al. The COVID-19 vaccine development landscape. Nat Rev Drug Discov. 2020;19(5):305-6.
8. Zhang C, Maruggi G, Shan H, Li J. Advances in mRNA Vaccines for Infectious Diseases. Front Immunol. 2019;10:594.
9. Prompetchara E, Ketloy C, Palaga T. Immune responses in COVID-19 and potential vaccines: Lessons learned from SARS and MERS epidemic. Asian Pac J Allergy Immunol. 2020;38(1):1-9.
*GlobalData search on 01-June-2020. GlobalData, John Carpenter House, UK.
Acknowledgments: Thank you to Celerion scientists Aernout van Haarst, Sumit Kar, Michelle Combs and Lorraine Rusch for editorial assistance.